
Podcast
MS-Selfie
Hosted by Gavin Giovannoni · EN
22episodes
Episodes
Newest firstAll episodes
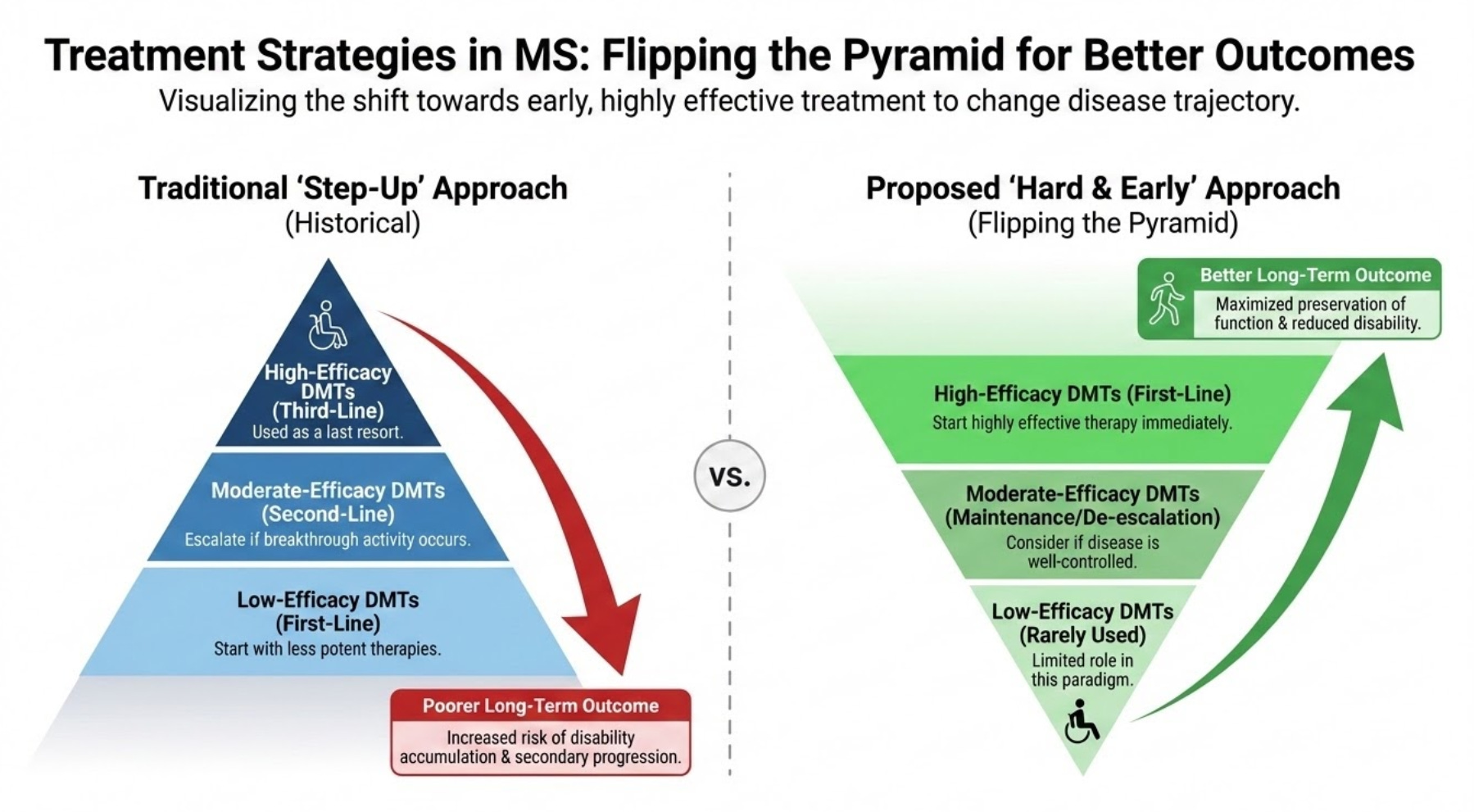
Should my MS be hit hard and early?
Apr 1500:03:53Tap to summarizeThis Swedish study below explains why I keep repeating myself, i.e. ‘flip the pyramid’ and ‘smouldering MS is the real MS’.The Swedes have investigated how different intensities of initial MS treatments impact long-term physical disability. They compared high-efficacy therapies with standard platform treatments in over 2,500 pwMS to distinguish worsening caused by relapses (relapse-associated worsening, or RAW) from progression that occurs independently of relapses (PIRA). They show that high-efficacy drugs significantly reduced the risk of relapse-associated worsening (RAW) and delayed reaching major disability milestones. However, neither treatment type showed a superior advantage in slowing progression independent of relapse activity (PIRA). This suggests that RAW and PIRA likely stem from different biological processes.These findings indicate that while modern DMTs excel at controlling inflammation (NEIDA), additional neuroprotective strategies are necessary to halt the disease’s gradual decline associated with SAW. The results of this study advocate for aggressive early intervention (hit-hard-and-early) and the development of therapies that target smouldering processes within the CNS and repair mechanisms.The findings of this study are not new and are in keeping with other published data. Sadly, however, there are likely too few patients treated with alemtuzumab and cladribine in this cohort and none treated with AHSCT. It is well known that the majority of pwMS on high efficacy treatments in Sweden are on off-label rituximab. I am told that Swedish neurologists now rarely use alemtuzumab, and the uptake of cladribine has also been slow. It would, however, be interesting to see if IRTs (immune reconstitution therapies), in particular alemtuzumab and AHSCT, made a difference in SAW. I say this because alemtuzumab and AHSCT are the most effective treatments we have for MS and have a profound impact on the end-organ in terms of reducing the rate of brain volume loss or brain atrophy. These two treatments do impact SAW, provided they are used early.It is becoming increasingly difficult to justify a low-efficacy platform DMTs approach when it is clear that pwMS treated with high-efficacy therapies first-line do so much better. Do you agree?A more interesting question for the next generation of neurologists to answer will be whether an induction-maintenance approach is superior to continued long-term immunosuppression, such as long-term anti-CD20 therapy. I note the Swedes are starting to test rituximab induction followed by cladribine as an exit strategy.The HIt HArd and hiT Early in Multiple Sclerosis Trial (HiHat) - ClinicalTrials.gov ID NCT07517185, Sponsor Uppsala UniversityI have been advocating this approach for several years, as it reduces the risks associated with long-term immunosuppression, allows you to recover your naive B-cell population for vaccinations, and, as cladribine is CNS-penetrant, targets CNS-resident B-cells and plasmablasts that are responsible for at least one component of smouldering MS. I have little doubt that this trial will work. Sadly, it is underpowered for efficacy, but who knows if the majority of subjects go into long-term remission, it may affect how both rituximab and cladribine are used in Sweden.If you are on long-term anti-CD20 therapy, would you consider switching to cladribine? Would you participate in a trial to test the safety and efficacy of anti-CD20 induction followed by an exit via cladribine?Guo et al. Long-term disability after initiation of platform versus high-efficacy disease-modifying therapy in relapsing-onset multiple sclerosis. J Neurol. 2026 Apr 1;273(4):240.Background: Several observational studies have compared high-efficacy and platform disease-modifying therapies (DMTs) with respect to long-term disability in relapsing-onset multiple sclerosis (MS), yet it remains unclear whether observed differences reflect relapse-associated worsening (RAW), progression independent of relapse activity (PIRA), or both.Methods: We included 2,563 DMT-naïve individuals with relapsing-onset MS enrolled in a population-based study linked to the Swedish MS registry (40 clinics, 2005-2019). The exposure was an initial DMT efficacy class (platform versus high-efficacy therapy), with platform as the reference. Cox models estimated hazard ratios (HRs) with 95% confidence intervals (CIs) for RAW, PIRA, and time to EDSS 3 and 4. EDSS trajectories were modelled using mixed-effects models. Follow-up started at DMT initiation and was censored at treatment switch, discontinuation, death, drop-out, or study end.Results: At treatment initiation, 1,987 participants started a platform DMT and 576 a high-efficacy DMT. High-efficacy therapy was associated with a lower risk of RAW (HR 0.60, 95% CI 0.38-0.92), while the risk of PIRA did not differ between treatment groups (HR 1.05, 95% CI 0.79-1.39). Risks of reaching EDSS 3 and EDSS 4 were also lower with high-efficacy DMT (EDSS 3: HR 0.26, 95% CI 0.17-0.38; EDSS 4: HR 0.32, 95% CI 0.18-0.54). EDSS trajectories increased more steeply among participants treated with the platform, with partial convergence toward the high-efficacy group over time.Conclusions: Our findings suggest that inflammatory and relapse-independent components of MS disability respond differently to current therapies and highlight the need for complementary neuroprotective strategies.Accidental readersIf you have been forwarded this email and are not an MS-Selfie subscriber, please consider subscribing and helping MS-Selfie expand its resources for the broader MS community. MS-Selfie relies on subscriptions to fund its curated MS-Selfie microsite, MS-Selfie books, MS-Selfie Infocards, and other activities that extend beyond the MS-Selfie Substack newsletters.Subscriptions and donationsMS-Selfie newsletters and access to the MS-Selfie microsite are free. In comparison, off-topic Q&A sessions are restricted to paying subscribers. Subscriptions are being used to run and maintain the MS Selfie microsite and other related activities, as I don’t have time to do this myself. You must be a paying subscriber to ask questions unrelated to the newsletters or podcasts. If you can’t afford to become a paying subscriber, please email a request for a complimentary subscription (ms-selfie@giovannoni.net).QuestionsIf you have questions unrelated to the newsletters or podcasts, please email them to ms-selfie@giovannoni.net. Prof. G will try to answer them as quickly as possible.Important Links📋 MS-Selfie microsite💰 Donations to MS-Selfie👈 Prof. G’s Backstory and CV💾 Prof. G’s MS Blog Archive❓ Conflicts of Interest🦋 BlueSky Social🔗 LinkedIn🖋 MediumGeneral DisclaimerPlease note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Queen Mary University of London or Barts Health NHS Trust. The advice is intended as general and should not be interpreted as personal clinical advice. If you have any problems, please tell your healthcare professional, who can help you. This is a public episode. If you'd like to discuss this with other subscribers or get access to bonus episodes, visit gavingiovannoni.substack.com/subscribe
Transcribe →
Q&A: managing advanced MS
Mar 1100:42:41Tap to summarizeIf you don’t have advanced MS, you may find the contents of this newsletter upsetting. It is not for everyone. Only read on if you want to know about managing advanced MS.QuestionI am at EDSS 8 and need to get my affairs in order. I cannot do anything really anymore, so is this the beginning of the final stages? I know you won’t sugarcoat the reply, so? I can no longer go to the toilet on my own and need to use the Peristeen system with my partner’s help. My partner uses a hoist to move me to and from bed/chair, etc. I have just had pneumonia, and the doctors said if I had a heart attack, they would not step in on medical grounds, even though I protested. What would you advise?NOTE: General Substack newsletters and the microsite are free; only Q&A sessions are restricted to paying subscribers. I can’t run and maintain the MS-Selfie microsite, so I must pay people to assist me. If people want to ask medical questions unrelated to the Newsletters or Podcasts, they either need to become paying subscribers or email (ms-selfie@giovannoni.net) to request a complimentary subscription.Prof G’s responseThank you for your questions and for being so honest about what you are going through. I get the sense that you are exhausted and anxious. I am sorry you are having to navigate such a heavy burden, especially after battling pneumonia and having such a distressing conversation with your doctors about end-of-life care. These conversations are best conducted well in advance, not during an acute crisis. This is why I urge all of you to complete an advance directive early on in your disease course and make sure your family knows about it and has a copy.You asked me not to sugarcoat things, so I will give it to you straight. Reaching EDSS 8 is a significant and difficult reality. It represents the advanced stages of physical disability in MS, which is why you are facing the loss of independence, needing the hoist, relying on the Peristeen system and your wife’s support. However, being in this advanced stage of disability does not necessarily mean you are in the final weeks or months of your life. Many people live for years and decades with an EDSS of 8. However, it does mean you are more vulnerable to complications, as evidenced by the flu.Given that vulnerability, you are right to get your affairs in order. Doing so is not a sign that you are giving up or that the end is near. It is simply about taking back some control, ensuring your exact wishes are legally documented, and giving you and your wife some peace of mind so you don’t have to worry about the administrative side later.Regarding the doctors telling you they would not step in if you had a heart attack or, by implication, another life-threatening event, I can only imagine how surprising or jarring and angering that was, especially as you protested. In the UK medical system, doctors can legally place a “Do Not Attempt Cardiopulmonary Resuscitation” decision on your file if they clinically believe the severe physical trauma of CPR would not be successful or would cause you more suffering due to your body’s current frailty and only if you and your family consent to it happening. However, your voice still matters. You are entirely within your rights to ask for a second medical opinion regarding this decision, to complete an advanced directive contrary to their advice and to demand a clearer conversation about your ongoing care plan going forward.Please try to be mindful of yourself. Your feelings and frustrations are valid, but anger will not help. Your neurological team should be there to support you through this. I would also recommend that they refer you to the palliative care team that has access to community services that can help you. For example, you would benefit from some form of counselling.As you are aware, from a treatment perspective, we don’t have any therapies to stop your disability from worsening or to reverse it. We think treatments such as cladribine may slow the rate of worsening, which is why we are conducting the CHARIOT-MS trial in advanced MS. We also know that comorbidities such as recurrent infections, pressure sores, poor metabolic health, poor sleep hygiene, low mood, etc., worsen MS. This is why you, your GP, and your local MS service need to focus on optimising the small things.I have looked after many people with advanced MS who have done this, and when the small things are looked after, they often notice an improvement in their quality of life.I hope this helps.I now realise the information I have given you is quite superficial. I have therefore produced a longer newsletter that covers topics you and others with more advanced MS may find helpful.Advanced MSAs you know, the EDSS ranges from 0.0, indicating a normal neurological examination, to 10.0, indicating death due to MS. The transition to an EDSS score of 8.0 represents a watershed in the clinical trajectory. At an EDSS of 8.0, the pwMS is usually restricted to a bed or a chair, or requires assistance in a wheelchair for mobility. While they may be out of bed for much of the day, their independent ambulatory capacity is usually curtailed, unless they have an electronically controlled wheelchair. At EDSS 8.0, pwMS generally retain effective use of their arms, with some preservation of many self-care functions. This clinical profile distinguishes them from pwMS at EDSS 8.5, who are restricted to bed for much of the day with only some effective arm use, and those at EDSS 9.0 or 9.5, who are totally confined to bed and face severe bulbar dysfunction, including problems with communication and swallowing.Because pwMS at EDSS 8.0 retain upper-extremity function and cognitive awareness, preserving their autonomy is a clinical priority and the reason we launched our #ThinkHand campaign almost a decade ago. We don’t think pwMS who are wheelchair users should be ignored. As part of the #ThinkHand campaign, we have been involved in designing and starting the O’HAND (ocrelizumab in PPMS), CHARIOT-MS (cladribine in advanced MS) and OCTOPUS trials. All these trials use the 9-hole peg test as the primary outcome measure or part of the composite endpoint.When you lose the use of your legs, your arms and hands function are essential for mobility, and explain why pwMS with advanced MS prioritise upper extremity function over lower limb function.At this stage of advanced MS, care must focus on supporting daily living and preventing complications (preventive neurology). The philosophy I promote relies heavily on the aggregation of marginal gains—the understanding that meticulous attention to seemingly “small things” yields improvements in the pwMS’s dignity, survival, and psychological well-being. Furthermore, the relationship between the pwMS and their carer(s) becomes a central focus of care. The psychological resilience, physical safety, and emotional well-being of the carer are inextricably linked to pwMS outcomes, necessitating support, respite strategies, and environmental adaptations to mitigate the burden of providing chronic care.ComorbiditiesThe management of a wheelchair-bound or bedbound pwMS does not occur in a vacuum. People living with advanced MS exhibit a higher incidence of comorbidities compared to the general population. The most prevalent conditions are high blood pressure, diabetes, heart disease, fibromyalgia, chronic lung disease, and mood disorders such as depression and anxiety.The presence of these comorbidities has been clinically linked to a faster worsening of disability, reduced overall quality of life, more frequent hospital admissions, and a regrettable shorter lifespan. Clinical studies indicate that pwMS who also suffer from vascular disease tend to require assistance with walking much sooner than those without vascular comorbidities. Therefore, the active management of metabolic syndrome and vascular health should not be peripheral to MS care, but a central part of it.Because a pwMS with EDSS 8.0 is often entirely sedentary, their risk for developing metabolic syndrome—a constellation of high blood pressure, elevated blood glucose, excess body fat around the waist, and abnormal cholesterol levels—increases due to a forced sedentary lifestyle. Maintaining a good relationship with your general practice or a primary care provider is, therefore, essential as most MS services don’t have the resources and expertise to manage these comorbidities.Respiratory functionRespiratory complications are recognised as one of the leading causes of death in pwMS, accounting for approximately 50% of all deaths in longitudinal studies. Emergency, unplanned, or non-elective admissions for respiratory crises in pwMS cost an average of £216,000 annually per NHS Clinical Commissioning Group (CCG), with £75,000 attributed to admissions for aspiration pneumonia. Prior to their dissolution, there were 106 CCGs in England; therefore, the cost to NHS England was ~£23M annually for this problem alone.The decline in respiratory function in MS is typically insidious and begins long before the pwMS reaches an EDSS of 8.0. Pulmonary function is affected even in the early stages of disability, primarily through weakening of core stabiliser muscles. This reduction in core strength translates into impaired pulmonary mechanics, diminished health-related quality of life, and worsening fatigue. However, the transition to a wheelchair-bound or bedbound state precipitates a much sharper decline in respiratory function. This manifests clinically as a weak and ineffective cough. When the expiratory muscles fail to generate sufficient force to clear the airway, and the inspiratory muscles are too weak to provide adequ...
Transcribe →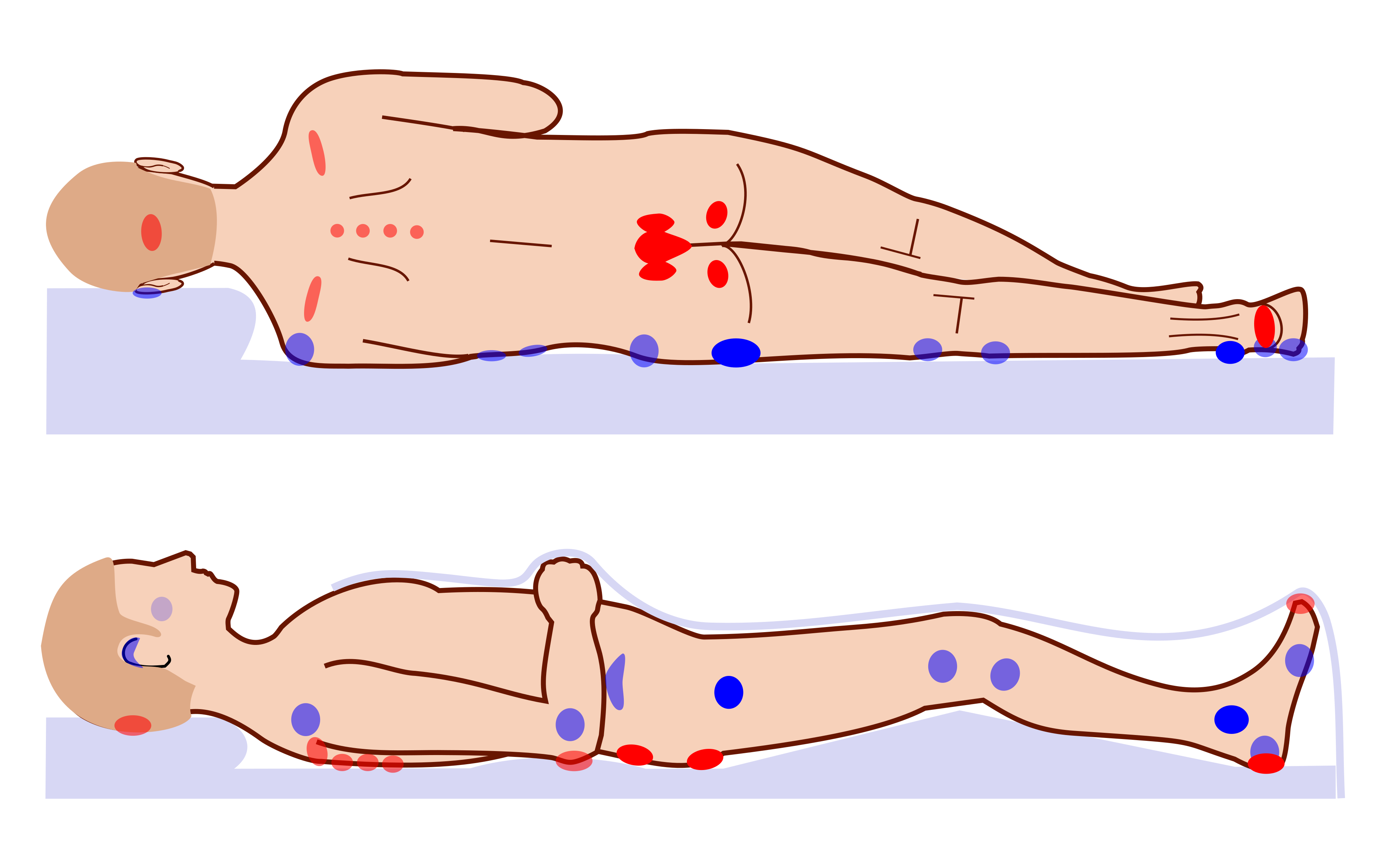
Pressure sores
Feb 2300:45:10Tap to summarizeI am sure many of you won’t want to know about the underbelly of living with advanced MS. As pwMS become more disabled, complications of being immobilised start to appear. The one that worries all HCPs working with patients who have MS is pressure sores or ulcers.Pressure ulcers remain a devastating clinical challenge encountered in the management of people with advanced MS. Pressure ulcers, pressure sores, bedsores, decubitus ulcers, or pressure injuries, are localised areas of cellular necrosis and tissue damage occurring in the skin and underlying soft tissues. They almost exclusively develop over bony prominences such as the sacrum, ischial tuberosities (hips), heels, and occiput (back of head). The following image from Wikipedia illustrates the most common sites for pressure ulcers. MS does not intrinsically or directly cause skin breakdown; however, motor and sensory loss, autonomic dysfunction, spasticity, and cognitive impairment associated with advanced MS are risk factors for pressure sores.The economic burden of pressure sores is very high and continues to escalate globally. In the UK, pressure ulcers rank among the top 10 harms in the NHS, affecting hundreds of thousands of patients annually. In 2004, pressure ulcers were estimated to cost the NHS between £1.4 billion and £2.4 billion per year, representing approximately 4% of total NHS expenditure at the time. More recent estimates indicate that the management of pressure ulcers costs the NHS over £3.8 million every single day. At an individual level, pressure ulcers diminish a patient’s health-related quality of life. The condition frequently confines individuals to strict bed rest, exacerbates social isolation, causes pain, and introduces the risk of life-threatening complications such as osteomyelitis and septicemia.I recall when I was a very junior neurology trainee looking after a young woman with MS and a large cavitation pressure ulcer over her sacrum. She spent more than 6 months on the ward having the pressure sore managed. Sadly, shortly after discharge, she developed recurrent septicaemia and passed away from infection. Whenever I see a patient with a pressure sore, I have flashbacks to my experience with this patient.The management of pressure ulcers within the MS population is complex and requires a multidisciplinary team to manage spasticity, pain and frequently immobility. Care pathways have shifted away from reactive wound care toward highly proactive, multidisciplinary prevention strategies integrated within frameworks like the National Wound Care Strategy Programme (NWCSP). The intersection of clinical care, patient safety, and legal accountability in the UK has transformed the reporting and management of severe pressure ulcers. Regulatory bodies, such as the Care Quality Commission (CQC), alongside legislative frameworks such as the Care Act 2014, have established notification and safeguarding protocols. These legal mechanisms are designed to determine whether severe tissue damage represents an unavoidable consequence of terminal clinical decline or a physical manifestation of systemic neglect and institutional failure. Yes, pressure sores are a notifiable condition under the NHS and must be investigated.Mechanisms of tissue failureThe aetiology of pressure ulcers in pwMS is multifactorial. The injury involves prolonged mechanical loading on soft tissues compressed between an external support surface—such as a mattress, wheelchair seat, or orthotic device—and an internal skeletal structure. The continuous pressure, friction, and shear forces are responsible for tissue injury. Under normal conditions, capillary closure pressure is approximately 32 mmHg. When external mechanical pressure exceeds this threshold for a sustained period, localised tissue perfusion is severely compromised. This results in hypoxia, the accumulation of metabolic waste products, and subsequent cellular necrosis. Vulnerable individuals lacking the capacity for spontaneous movement, irreversible ischaemic damage can commence in as little as 20 minutes.Shear forces represent a particularly insidious and destructive mechanism. Shearing occurs when the epidermal layer remains stationary against a support surface, anchored by friction, while the underlying skeletal structures and deep fascial layers shift or slide. This dynamic is frequently observed when a patient with poor core control slides down the incline of a hospital bed or assumes a slumped posture within a wheelchair. This parallel, opposing force severely distorts, stretches, and ultimately ruptures deep fascial blood vessels, initiating ischaemia from the deep muscle tissues outward toward the skin. This mechanism frequently results in suspected deep tissue injuries that cause massive subdermal necrosis while initially presenting only as intact, discoloured skin on the epidermal surface. Friction, conversely, acts directly on the epidermal layer. Functioning like sandpaper, friction physically strips away the protective stratum corneum during transfers or spasms, drastically lowering the tissue’s threshold for pressure-induced necrosis and exposing deeper layers to bacterial colonisation.While these mechanical forces are the direct forerunners of ulceration, the intrinsic neurological deficits imposed by MS render the tissues susceptible to injury, stripping away the body’s natural defensive mechanisms. Advanced MS is usually associated with paraparesis or quadriparesis, which is associated with a lack of spontaneous micro-movements and weight-shifting behaviours that healthy individuals continuously utilise to redistribute weight and permit capillary reperfusion during sitting or sleeping. At the same time, damage to the ascending sensory pathways results in significant hypoaesthesia or complete anaesthesia. The loss of protective pain feedback, which would normally alert the brain to impending tissue ischaemia and a change in posture, is reduced or lost in advanced MS.Spasticity (hypertonia), muscle spasms, and joint contractures alter resting posture, often forcing limbs into unnatural and asymmetrical positions, and create friction and shear forces as the patient’s body rubs against support surfaces during movement of spasms.Comorbid bladder and bowel dysfunction leads to frequent urinary and faecal incontinence. Prolonged exposure to moisture, along with bacterial and fungal overgrowth, macerates the epidermis. Moisture accelerates frictional damage and introduces the complicating factor of moisture-associated skin damage (MASD), which frequently co-occurs with pressure ulceration.Advanced MS is often accompanied by dysphagia, fatigue, and severe depression, all of which contribute to reduced caloric and protein intake. This leads to malnutrition and low albumin levels. Skin integrity is intrinsically linked to nutritional status; malnourished, hypoalbuminaemic tissues lack the amino acid substrates required for repair, collagen synthesis, and maintenance of interstitial oncotic pressure, resulting in oedema and diminished resistance to hypoxic stress.EpidemiologyPressure ulcers are a pervasive, escalating global health crisis, exacerbated by ageing populations, increasingly fragmented care networks, and the rising prevalence of complex chronic diseases. In the Global Burden of Disease (GBD) Study, report that the incident cases of pressure ulcers more than doubled from 1,142,594.8 in 1990 to 2,468,317.5 in 2021, and the resultant mortality attributed to pressure ulcers rose from 16,741.1 to 37,032.7 globally, and DALYs surged from 408,887.0 to 803,747.4 over the same period.Despite these increases in absolute burden driven by population growth and ageing, the age-standardised incidence rate (ASIR) decreased slightly, indicating that, while the total number of patients suffering is higher, the per-capita risk, when adjusted for age, has stabilised or marginally improved due to preventive measures.Sadly, the burden of pressure ulcers correlates with the socio-demographic index (SDI), with significantly higher burdens observed in elderly females and a rising incidence in elderly males. In a prospective cohort study of 185 pwMS without existing skin breakdown, the incidence rate of developing a new pressure ulcer was documented at 10.8% (20 patients) (see paper below). The development of pressure ulcers was not random; predictors of tissue failure included a history of previous stroke, being bed- or chair-bound, poor nutrition and urinary or faecal incontinence.In the UK, pressure ulcer prevalence in highly dependent populations ranges from 11 to 13%. However, when looking strictly at wheelchair users—who constitute a large and vulnerable proportion of patients with advanced MS—registry studies indicate varying prevalence rates heavily dependent on the underlying neurological pathology. Among young wheelchair users overall, 55% experience at least one pressure ulcer a decade post-injury or disease onset. Specifically within the MS sub-population of chronic wheelchair users, the cross-sectional prevalence of current pressure ulcers is estimated at 4%.Within intensive care units (ICUs) and orthopaedic surgical wards where MS patients may be admitted during severe disease relapses or for unrelated acute pathologies, incidence rates peak dramatically. General ICU prevalence averages 6.8%, with an incidence rate of 3.8%. Orthopaedic surgery wards have alarmingly high incidence rates of 18.5%, emphasising the danger of prolonged surgical immobilisation. The sacrum is overwhelmingly the most affected area, accounting for up to 68% of ulcers, followed closely by the heels, left and right trochanters, and ankles. When analysing severity across generalised hospital populations, stage 1 and stage 2 ulcers account for the vas...
Transcribe →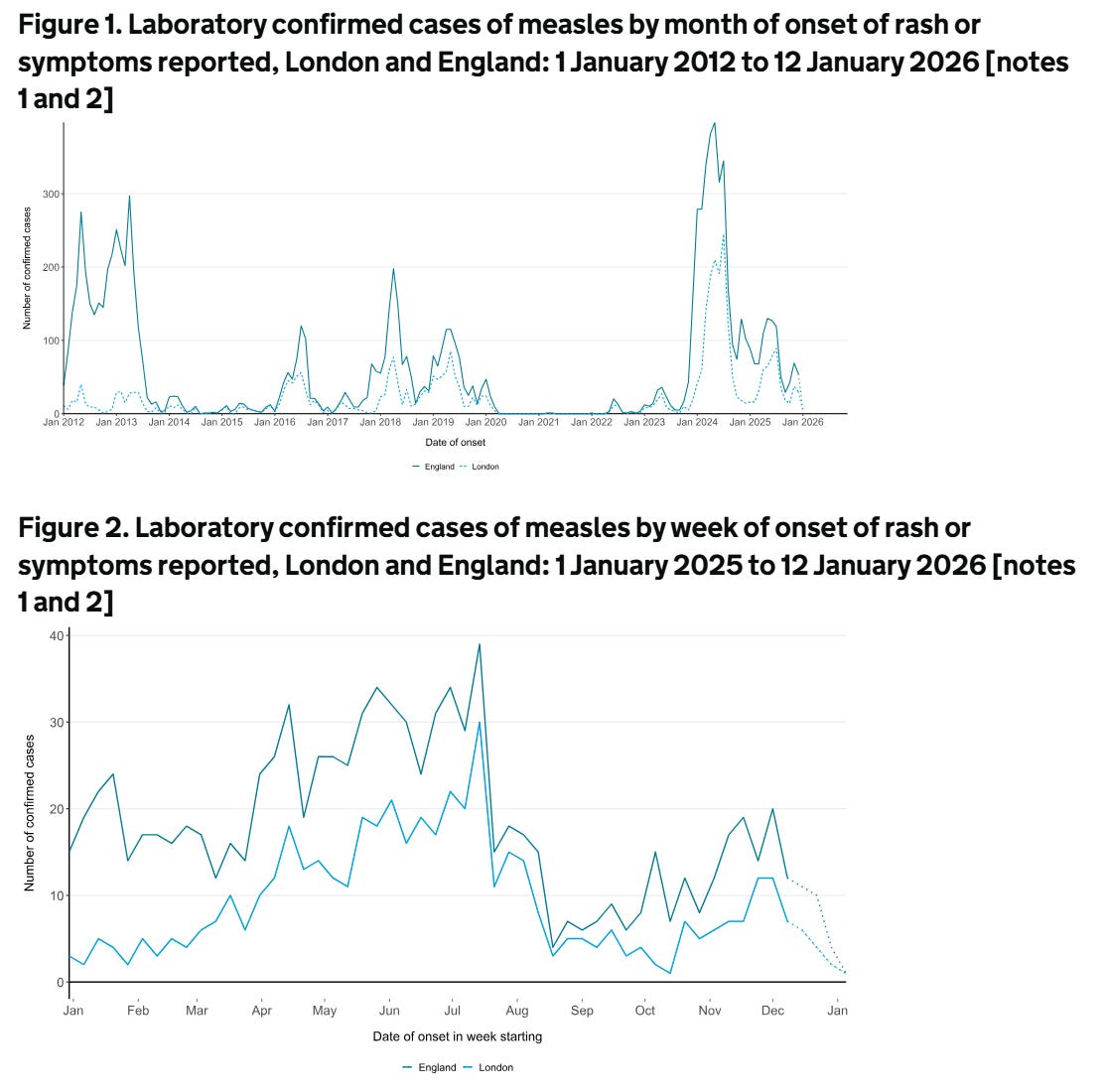
Should I be worried about measles?
Jan 2900:08:44Tap to summarizeIf you have multiple sclerosis (MS) and are on chronic immunosuppression, you need to check your medical records to make sure you have had the measles vaccine as a child. Due to vaccine hesitancy, MMR (measles, mumps, and rubella) vaccine uptake has dropped considerably since the late 90’s and noughties. As a result, many countries now have both endemic and epidemic levels of measles infections. This is clearly very worrying. The bad news is that the World Health Organisation (WHO) has stated that the UK is no longer considered to have eliminated measles. The WHO has said measles transmission was re-established in the UK in 2024, following a plateau in vaccination coverage and a surge in cases. A total of 3681 measles cases were recorded in England in 2024. This will underestimate the true incidence of measles, as these figures rely on accurate diagnoses and on cases being reported to the Health Security Agency. Now, let's say you are a person with MS who did not have the MMR vaccine because your parents were worried about the safety of the MMR vaccine. You would now be vulnerable to getting infected as an adult. In the past, you could have relied on herd immunity, i.e. all the people around you who had the vaccine and are immune to measles would prevent the virus from spreading in the community and protect you. Unfortunately, herd immunity is simply not good enough. The latest figures from the UK’s Health Security Agency for England show that in 2024-2025, 91.9% of five-year-olds had received one dose of the MMR vaccine, which is largely unchanged from 2023-2024 and the lowest level since 2010-2011. Just 83.7% of 5-year-olds had received both MMR doses, down year-on-year from 83.9% and the lowest level since 2009-2010. The WHO recommends that at least 95% of children receive each vaccine to achieve herd immunity.My worry is that people with MS will get measles, and because they are on an immunosuppressive therapy, will get complications, in particular encephalitis.Measles encephalitisMeasles encephalitis is a serious complication of the measles virus infection that often leads to permanent brain damage or death. While measles is often thought of as a respiratory illness with a rash, the virus can infect the central nervous system. This complication occurs in approximately 1 in every 1,000 people who get measles, but it is much more common in people who are immunocompromised.Acute measles encephalitis usually occurs during or shortly after the initial infection (often whilst the rash is still present). It is often caused by the immune system attacking the brain (an autoimmune reaction) rather than the virus directly attacking brain tissue, though direct viral invasion also happens. It is associated with a high fever, headache, stiff neck, vomiting, drowsiness, seizures, and coma. It typically begins about 6 days after the rash onset. The mortality rate is approximately 10–15%. Among survivors, about 25% suffer permanent neurological damage, such as intellectual disability, seizures, or deafness.Subacute Sclerosing Panencephalitis (SSPE) is a rare, chronic, and fatal form of measles encephalitis. It is caused by a persistent measles virus infection in the brain that reactivates years later. It typically develops 7 to 10 years after the person has recovered from the initial measles infection. It starts with subtle behavioural changes and memory loss, progressing to muscle spasms (myoclonus), seizures, and eventually a vegetative state. It occurs in roughly 1 in 10,000 to 1 in 100,000 people who had measles, but the risk is higher for those infected with measles before the age of two.Measles inclusion body encephalitis (MIBE) is a specific form found in people with compromised immune systems (e.g., those with HIV, organ transplants or on immunosuppressive treatments). It usually occurs a few months after the initial measles infection. It is usually fatal, with death often occurring within weeks of symptom onset.There is no specific antiviral treatment for these forms of measles encephalitis once they have begun; healthcare professionals can only provide supportive care (hydration, seizure management, etc.) to help the body fight the infection. Therefore, the only effective protection is prevention via the MMR vaccine. The vaccine prevents the initial measles infection, thereby eliminating the risk of these neurological complications.It is only a matter of time before a person with MS who has not received the MMR vaccine gets measles and one of the forms of measles encephalitis. I am particularly concerned about pwMS on natalizumab who will get a subacute infectious syndrome that may mimic PML or a prion disease such as Creutzfeldt-Jakob disease. PwMS on anti-CD20 therapies and S1P modulators are also at risk. PwMS treated with immune reconstitution therapies and have reconstituted their immune systems should be okay, as they can mount an immune response to the virus.Measles hyperimmune immunoglobulin (or human normal immunoglobulin - HNIG) is used as post-exposure prophylaxis (PEP) for susceptible individuals at high risk of severe complications from measles. It provides immediate, temporary antibodies and is recommended when the MMR vaccine cannot be used or when exposure has occurred in highly vulnerable people. The eligible groups for immunoglobulin (HNIG/IVIG) are individuals who have had significant exposure to a confirmed case of measles, are not immune (no history of 2 doses of vaccine or measles disease), and are within the 6-day exposure window. This includes infants under 1 year of age, pregnant women and immunocompromised individuals (e.g., bone marrow transplant recipients, HIV/AIDS patients, those on high-dose immunosuppressive therapy). IVIG is usually recommended for immunosuppressed individuals, while intramuscular HNIG is used for pregnant women and infants. For maximum effectiveness, immunoglobulin should be administered as soon as possible after exposure, ideally within 72 hours, but it can be given up to 6 days after exposure. It is typically administered via intramuscular injection or intravenously.HNIG/IVIG is not a replacement for vaccination. Individuals who receive HNIG/IVIG must still receive the MMR vaccine once they are no longer immunocompromised, ideally at least 6–8 months after the immunoglobulin injection. The latter is to wait for the anti-measles IgG from HNIG/IVIG to be cleared from the body, so it won’t neutralise the vaccine.Please note that, on the NHS, approval for measles immunoglobulin treatment is generally provided by a consultant in health protection or a virologist, not by a neurologist. The latter can delay things. I speak from personal experience as I have had one patient on anti-CD20 therapy who has a large family, and none of his children have had the MMR vaccine. The reasons for not vaccinating his children are complex and related to religion and vaccine scepticism. However, when one of his children developed measles, we had to activate this pathway. Thankfully, urgent MMR serology showed he must have had the vaccine as a child. He was not born in the UK and had no documentation of his vaccine status.Please note that it is now part of our standard practice to check all patients’ MMR status before starting chronic immunosuppressive therapy. Since starting this practice, we have identified a small number of people who have been MMR negative who have then had to have the MMR vaccine prior to initiating treatment. The problem is that once you are on an immunosuppressive therapy, you can’t have the vaccine, as it is a live vaccine. This is one of the arguments in favour of IRTs (immune reconstitution therapies) for treating MS. Once your immune system has reconstituted after cladribine, alemtuzumab or AHSCT, live vaccines are relatively safe and not contraindicated.Do you know your MMR status? If not, you may want to know particularly if you are about to start an immunosuppressive therapy. At the same time, please be vigilant for measles in your area, and if you come into contact with someone with measles, let your HCP know.Have any of you not had the MMR vaccine? Have you recently come into contact with someone with measles?Accidental readersIf you have been forwarded this email and are not an MS-Selfie subscriber, please consider subscribing and helping MS-Selfie expand its resources for the broader MS community. MS-Selfie relies on subscriptions to fund its curated MS-Selfie microsite, MS-Selfie books, MS-Selfie Infocards, and other activities that extend beyond the MS-Selfie Substack newsletters.Subscriptions and donationsMS-Selfie newsletters and access to the MS-Selfie microsite are free. In comparison, off-topic Q&A sessions are restricted to paying subscribers. Subscriptions are being used to run and maintain the MS Selfie microsite and other related activities, as I don’t have time to do this myself. You must be a paying subscriber to ask questions unrelated to the newsletters or podcasts. If you can’t afford to become a paying subscriber, please email a request for a complimentary subscription (ms-selfie@giovannoni.net).QuestionsIf you have questions unrelated to the newsletters or podcasts, please email them to <a target="_blank" href="mail...
Transcribe →
Male and female-specific MS care pathways
Jan 2000:23:14Tap to summarizeIn response to comments from yesterday’s podcast and newsletter, I have prepared a more detailed analysis of the issues supporting gender-specific MS services. I hope this clarifies things better. Historically, the clinical and epidemiological descriptions of MS have stressed differences between women and men with MS. This is often referred to as sexual dimorphism. MS is a disease that disproportionately affects women, with current prevalence estimates indicating a female-to-male ratio of approximately 3:1. This ratio has widened over the past century, largely attributed to environmental shifts—such as smoking, obesity, and vitamin D deficiency—that appear to impact female susceptibility more than male susceptibility. However, these changes in risk factors only explain a small proportion of the increased incidence and prevalence of MS in women. Consequently, the “typical” MS patient is often conceptualised as a young woman of childbearing age. This ‘heuristic’ has fundamentally shaped the design of clinical services, support networks, and therapeutic monitoring protocols in clinical practice.However, this statistical predominance masks clinical reality: while women are more susceptible to acquiring the disease, men often experience a more severe, rapid, and neurodegenerative course. The “male paradox” in MS—lower susceptibility but higher severity—challenges the uniform application of clinical guidelines. Standardised care pathways, such as those outlined by the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) and the American Academy of Neurology (AAN), largely advocate a generalised approach to pharmacological management and symptom monitoring, with sex-based differentiation primarily in reproductive safety.I think it is time to challenge the “one-size-fits-all” model. There is data on genetic, hormonal, and immunological differences that warrant this. Alongside clinical registry data and emerging clinical pilots suggest that the care of women and men with MS differs sufficiently to warrant distinct service based on gender. Biological sex is not merely a demographic variable but a biological determinant of disease pathology, necessitating a shift from gender-neutral to gender-responsive care. This could be part of precision medicine, i.e., tailoring care to the individual.Biological determinantsTo determine whether clinical care pathways should diverge, one must first establish whether the underlying biological substrates of the disease differ significantly between the sexes.While the HLA complex remains the main genetic risk locus for both sexes, sex-specific genetic factors modulate this risk. The “Four Core Genotypes” (FCG) mouse model aims to disentangle the effects of sex chromosomes from those of sex hormones. Utilising this model indicates that the sex chromosome exerts an independent effect on neurodegeneration; i.e., the presence of an XY chromosomal complement is associated with greater clinical severity and increased neuropathology compared to an XX complement, even when hormonal environments are controlled. This indicates that males may possess an intrinsic genetic vulnerability to neurodegenerative processes.Specific gene polymorphisms exhibit sex-dependent risk profiles. An example is the galanin gene (GAL), which codes for a neuropeptide widely expressed in the brain and spinal cord. Variants in the GAL gene have been associated with MS susceptibility and delayed onset specifically in men, whereas in women these variants appear to influence disease progression rather than susceptibility. This implies that the genetic “motor” driving disease onset in men may differ from that in women. Whether different therapeutic targets are needed in men remains to be determined.Neuroendocrine factors: oestrogens vs. androgensSex hormones—oestrogens and progestogens in females, and androgens in males—are immunomodulators that influence the blood-brain barrier (BBB) permeability, immune cell differentiation, and remyelination efficiency. Women possess a more robust adaptive immune system than men, characterised by stronger humoral (antibody-mediated) and Th1 (pro-inflammatory) responses. This evolutionary trait, while beneficial for fighting infection, may lower the threshold for developing autoimmune disease. Oestrogens typically enhance immune reactivity, which may contribute to the higher incidence of relapsing-remitting MS (RRMS) in women.However, the role of oestrogens is biphasic and dose-dependent. At physiological levels present during the menstrual cycle, oestrogens may facilitate inflammation. Conversely, at the high concentrations observed during pregnancy (particularly oestriol), they exert immunosuppressive and neuroprotective effects, which are believed to be responsible for inducing temporary remission. In comparison, the postpartum period represents a withdrawal of this protection, leading to a rebound in relapse rates. The cessation of ovarian function at menopause is associated with an acceleration of disability accumulation, suggesting that oestrogens provide a “neuroprotective buffer” that delays the onset of secondary progression.Testosterone acts as a natural immunosuppressant, suppressing Th1 differentiation and promoting a shift toward the anti-inflammatory Th2 phenotype. This mechanism may protect many men from developing MS. However, once the disease is established, men face a more rapid decline, which may be due to a failure of androgen-mediated repair.Men with MS frequently exhibit lower testosterone levels than age-matched healthy controls. This hypogonadal state is not merely a comorbidity but appears to be a driver of pathology. Testosterone is essential for maintaining muscle mass, cognitive function, and synaptic plasticity. In the context of MS, low testosterone correlates with increased cognitive fatigue, reduced grey matter volume, and poorer functional outcomes. The vulnerability of male oligodendrocytes to oxidative stress and excitotoxicity, combined with lower levels of neuroprotective androgens, creates an environment for rapid neurodegeneration.NeuropathologyWhile women tend to exhibit a disease course dominated by peripheral inflammation (relapses and gadolinium-enhancing lesions) during their reproductive years, men exhibit a predominance of neurodegenerative markers earlier in the disease course. Male patients accumulate “smouldering” or chronic active lesions—characterised by iron-laden rims of activated microglia—at a higher rate than women.MRI studies demonstrate that men exhibit more rapid whole-brain and grey-matter atrophy, particularly in deep grey-matter structures such as the thalamus and cortex. This correlates with the faster cognitive decline observed in male cohorts.Animal models suggest that remyelination efficiency is superior in females, potentially due to oestrogen’s influence on oligodendrocyte precursor cells. Male oligodendrocytes appear more vulnerable to metabolic stress, leading to permanent axonal loss rather than repair.Prognostic disparitiesMen are typically diagnosed at a later age than women and are significantly more likely to present with primary progressive MS (PPMS). Registry data indicate that while relapsing-remitting MS (RRMS) is the most common form for both sexes, the proportion of men with PPMS is significantly higher (approximately 22% vs. 8% in women). Even among those with relapse-onset disease, men convert to secondary progressive MS (SPMS) at a younger age and after a shorter disease duration than women.4This diagnostic latency in men is multifactorial. Biologically, the higher threshold for inflammatory symptoms (relapses) in men may mask the disease until structural damage reaches a critical threshold. The perception of MS as a “woman’s disease” may delay clinical suspicion in men presenting with non-specific motor or cognitive symptoms, leading to diagnostic delays. Natural history studies confirm that male sex is an independent predictor of poor prognosis. Interestingly, the sex difference in relapse rates disappears after age 50—roughly the age of menopause.ComorbiditiesThe comorbidity profile also differs, with some comorbidities affecting survival and disability differently. Men with MS have higher rates of cardiovascular and metabolic comorbidities, including diabetes, hypertension, and ischemic heart disease. These conditions act synergistically with MS pathology. Hypertension and diabetes impair microvascular perfusion in the CNS, reducing the metabolic reserve of neurons already under attack by the immune system. This double hit accelerates brain atrophy and disability worsening in men. Conversely, women with MS report higher rates of other autoimmune conditions (e.g., thyroid disease, psoriasis) and anxiety/depression. While these impact quality of life, they do not typically accelerate neurodegeneration at the same rate as vascular comorbidities.Mortality and suicide riskMortality risks also differ significantly. While MS reduces life expectancy for all patients, the standardised mortality ratio (SMR) and suicide risk show profound gender imbalance. Men with MS have a higher rate of completed suicide compared to women, despite women reporting higher rates of suicidal ideation and attempts. This is likely due to the suicide methods chosen by men and the psychological strain of physical dependency, which conflicts with traditional masculine roles. The loss of physical autonomy strikes at the core of the male provider/protector role, creating a distinct psychological crisis for men that general depressi...
Transcribe →
Gender inequality in MS care
Jan 1900:06:09Tap to summarizeLast week, a colleague sent me the publication below (van Laar et al., 2024).The publication highlights that while gender equality efforts traditionally focus on women, including men is essential for achieving lasting social progress. The authors argue that restrictive gender roles significantly damage men’s physical and mental health by discouraging help-seeking and rewarding risky behaviours. Do you agree? Furthermore, men often perceive diversity and inclusion initiatives as a zero-sum threat to their status, leading to resistance or psychological blindness toward their own privilege. Again, do you agree? In the workplace, masculinity contests create competitive environments that harm well-being for all employees and prevent men from engaging in care-oriented roles. To move forward, the authors suggest mobilising men as allies by raising awareness of how equality benefits them personally. Ultimately, true freedom from gender stereotypes requires a holistic approach that allows every individual to reach their potential regardless of traditional norms.Call to actionMy colleague suggested doing two things for men with MS. Firstly, improving care and counselling across gender/sex concordance in MS care. In practice, that would be offering men with MS male doctors and women with MS female doctors. This is easier said than done, particularly in healthcare systems with few resources. I am aware of many centres in the UK where the neurology consultants covering the MS service are of the same sex.Another suggestion is to improve benchmarks and guidelines for comprehensive, lifespan care for men. For example, young men with MS tend to have evolving mental health needs different to those of young women. In midlife, men with MS may need more support for sexual dysfunction, fertility and family planning. And finally, in older men with MS, ageing mechanisms are different, as well as the mix of comorbidities. Men don’t have a biological menopause that is defined and managed by the medical system. There is no male HRT. Men are susceptible to prostatism that needs to be investigated and managed differently from bladder dysfunction in women.Some obvious questions come to mind:* Are men with MS managed and treated differently from women with MS in your centre or country?* The epidemiology of MS differs between men and women. Do we need to explain this to men?* What male-specific issues in MS management differ compared to women?* Do men with MS need a different management pathway, counselling, treatments, etc.?* Are there cultural issues around how men with MS interact and use healthcare that can affect their management and outcomes?* What male-related research priorities need addressing?* Is there any evidence that males with MS are neglected relative to their female counterparts?I would be interested in your take on the issues raised in this paper and the questions I have proposed. I have suggested branding MS a pink-ribbon disease in the past. Maybe we shouldn’t forget that it is also a light-blue ribbon disease.PaperVan Laar C, Van Rossum A, Kosakowska-Berezecka N, Bongiorno R, Block K. MANdatory - why men need (and are needed for) gender equality progress. Front Psychol. 2024 Mar 1;15:1263313.Some criticisms of the paperI found it interesting that all the authors on the paper were women. They identify several limitations to their approach and highlight potential criticisms of men's involvement in gender equality efforts. These criticisms generally fall into categories regarding the scope of the research, the potential for “backfire” effects when involving men, and the risks of performative support.Their review relies heavily on a “western binary view of gender,” where individuals are defined strictly as men or women. The authors admit this approach excludes the experiences of non-binary and gender-fluid individuals and does not reflect how a growing number of people define themselves, though they argue this focus is necessary to understand how to remove the specific power of traditional gender stereotypes. I know this is a woke issue, but it needs to be said.The majority of the research reviewed comes from “WEIRD” countries (Western, Educated, Independent, Rich, and Democratic). Consequently, the findings will not fully account for cultural, ethnic, religious, or national differences in how men experience gender roles. They discuss men as a single group, which risks overlooking their heterogeneity. I make the point regularly that fathers with daughters are different to men without daughters; we simply have a different worldview of women.Many men (due to ethnicity, social class, physical ability, or sexual orientation) do not possess the same levels of privilege usually associated with men in WEIRD countries.Engaging with men can potentially reproduce dominance or paternalistic relationships. For example, men may dominate interactions in gender-equality groups or claim “expert knowledge in areas they know little about”. Furthermore, specific forms of protest, such as men walking in high heels, can sometimes be derisive and reinforce rather than challenge gender inequalities. A criticism of involving men is that they often receive more recognition for their advocacy than women do. Men may be perceived as more credible and less self-interested, which can generate resentment among women and highlight the very inequality the movement seeks to dismantle.Strategies to include men can sometimes decrease women’s engagement. The authors cite research showing that explicitly inviting men to participate in gender-parity task forces led to fewer women volunteering. Additionally, when men take leadership roles in protests (rather than supportive roles), it can reduce women’s identification with the movement. There is also a risk of “performative allyship” or “lip service,” in which men engage in easy, costless actions to boost their reputations without a genuine commitment to change. This appearance of change can ultimately cost the movement by replacing actual progress.While the paper advocates appealing to men’s group-based interests (e.g., better health, better relationships), the authors note that this strategy can be counter-productive. It risks normalising the idea that men should only engage when they stand to “visibly benefit,” potentially hindering progress in areas where achieving equality requires men to give up power or privilege.Accidental readersIf you have been forwarded this email and are not an MS-Selfie subscriber, please consider subscribing and helping MS-Selfie expand its resources for the broader MS community. MS-Selfie relies on subscriptions to fund its curated MS-Selfie microsite, MS-Selfie books, MS-Selfie Infocards, and other activities that extend beyond the MS-Selfie Substack newsletters.Subscriptions and donationsMS-Selfie newsletters and access to the MS-Selfie microsite are free. In comparison, off-topic Q&A sessions are restricted to paying subscribers. Subscriptions are being used to run and maintain the MS Selfie microsite and other related activities, as I don’t have time to do this myself. You must be a paying subscriber to ask questions unrelated to the newsletters or podcasts. If you can’t afford to become a paying subscriber, please email a request for a complimentary subscription (ms-selfie@giovannoni.net).QuestionsIf you have questions unrelated to the newsletters or podcasts, please email them to ms-selfie@giovannoni.net. Prof. G will try to answer them as quickly as possible.Important Links📋 MS-Selfie microsite💰 Donations to MS-Selfie👈 Prof. G’s Backstory and CV💾 Prof. G’s MS Blog Archive❓ Conflicts of Interest🦋 BlueSky Social🔗 LinkedIn🖋 MediumGeneral DisclaimerPlease note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Queen Mary University of London or Barts Health NHS Trust. The advice is intended as general and should not be interpreted as personal clinical advice. If you have any problems, please tell...
Transcribe →
Q&A 139 - tolebrutinib and the FDA
Jan 1500:17:18Tap to summarizeQuestionProf G, could you do an article on what is going on with tolebrutinib in the States? Having a hard time processing the FDA rejection letter. Not sure what this means for the field.NOTE: General Substack newsletters and the microsite are free; only Q&A sessions are restricted to paying subscribers. I can’t run and maintain the MS-Selfie microsite, so I must pay people to assist me. If people want to ask medical questions unrelated to the Newsletters or Podcasts, they either need to become paying subscribers or email (ms-selfie@giovannoni.net) to request a complimentary subscription.Prof G’s responseYou can download the FDA response here. This is the informing Genzyme Corporation (Sanofi) that its application for the tolebrutinib cannot be approved in its current form. The primary obstacle to authorisation is a significant risk of severe liver injury, which the FDA believes outweighs the potential clinical benefits.Despite Sanofi’s attempts to manage this toxicity through intensive monitoring, the FDA identified several “life-threatening cases” during clinical trials. Additionally, the FDA noted substantial uncertainty regarding the drug’s effectiveness across multiple sclerosis subpopulations, particularly in those with inactive disease. To move forward, Sanofi must provide more robust safety data and potentially identify a specific patient group or groups where the therapeutic advantages clearly justify the high risks.The most critical factor in the FDA’s decision was the risk of severe drug-induced liver injury (DILI). The FDA characterised this risk as “substantial and unusually high” for drug development programmes in general, and specifically for MS therapies. In the Phase 3 development programme, there were 6 cases meeting Hy’s Law criteria (indicators of severe hepatotoxicity) out of approximately 2,700 subjects, including one patient who required a liver transplant and subsequently died.The FDA noted that even a single Hy’s Law case is a signal of high hepatotoxicity, and most drugs withdrawn from the market for this reason have rates of severe injury far lower than what was observed with tolebrutinib. While DILI is a known class effect of BTK inhibitors, tolebrutinib’s risk of fatal DILI appears to be “among the highest in the class” and exceeds the risk observed with all other approved MS therapies.The FDA concluded that the proposed Risk Evaluation and Mitigation Strategy (REMS) was insufficient. Although Sanofi implemented weekly liver monitoring, severe liver injury cases continued to occur even under this strict protocol. Consequently, the Agency determined that the benefit-risk assessment must assume severe and potentially fatal DILI cases would occur in a postmarketing setting regardless of monitoring requirements.The FDA identified significant issues with the efficacy data supporting the proposed indication for non-relapsing secondary progressive MS (nrSPMS). The study population was heterogeneous, containing both “active” and “non-active” SPMS patients, which complicated the assessment of benefit. The treatment effect was largely driven by a small subgroup (13% of enrolled subjects) with baseline gadolinium-enhancing lesions, indicating active SPMS. While the drug showed efficacy here, there are already approved therapies for active SPMS that do not carry the same magnitude of liver risk. Therefore, the benefits in this group were not anticipated to outweigh the severe DILI risk.For patients without baseline enhancing lesions—the population with the greatest unmet need—the treatment effect was significantly smaller and uncertain. The FDA stated that therapies with greater risks require a “greater magnitude and certainty of benefit” to support approval. The potential benefit in this population was deemed inadequate to justify the severe liver risks.Their analysis of the data showed that the treatment effect was substantially diminished in patients who had previously tried two or more prior MS therapies. The FDA rejected Sanofi’s claim regarding “slowing disability accumulation independent of relapse activity”. The Agency cited several reasons for this rejection. The concept lacks widely accepted criteria and relies on post hoc analyses. There is limited understanding of whether BTK inhibitors address the underlying pathophysiology of progression. They also highlighted a separate study in primary progressive MS that has recently failed to demonstrate a benefit on its primary endpoint of confirmed disability progression, further undermining the mechanistic rationale for this claim (see Tolebrutinib PPMS trial is negative, 15-Dec-2025). I would counter the latter argument that the PPM population in the Perseus trial differs significantly from the nrSPMS population in the Hercules trial. Ultimately, the FDA could not identify a clinically identifiable population for whom the benefits of tolebrutinib outweighed the “serious and unusually high risk of severe DILI”.My initial thoughtsThis is not the first or last time this has happened with MS DMTs and the FDA. They did the same with natalizumab (Tysabri), alemtuzumab (Lemtrada), and cladribine (Mavenclad) based on PML, secondary autoimmunity & other adverse events (infusion reactions, infections, …) and cancer risk with each DMT, respectively. All these drugs eventually got to market. I am confident there is a way forward.Defining a more responsive subgroupThe FDA is clearly worried about giving tolebrutinib a wide label for smouldering MS. This is because when you look for it using sensitive measures, for example, cognitive and other neurological stress tests, the majority of people with MS have smouldering disease. This means that the use of tolebrutinib would likely spread like wildfire to most subgroups of MS, and hence, the risk of life-threatening liver toxicity would increase. The FDA determined that, in its present form, a favourable benefit-risk profile could not be established for any patient subpopulation. The challenge for Sanofi is to identify a subgroup that would benefit most from the drug.Interestingly, in a post-hoc analysis, subjects with early-relapsing MS who had paramagnetic rim lesions (PRLs) responded better to tolebrutinib than those without PRLs. We have yet to see the same data for the PPMS cohort. But if PRLs serve as a prognostic marker in this population, it would make a compelling case for using this biomarker.Please see the MS-Selfie newsletter on ACTRIMS 2025 Highlights (3-Mar-2026), which covers this analysis.As you are aware, PRLs are one biomarker of smouldering MS lesions, and their presence is associated with poorer outcomes. PRLs are considered a “red flag” in MS because they represent chronic, smouldering inflammation that actively destroys brain tissue long after the initial attack has passed. While a typical MS lesion stabilises and “heals”, PRLs behave like a slow-burning fire that continues to expand outward, causing progressive damage.The defining feature of a PRL is a dark ring seen on specific MRI scans (susceptibility-weighted imaging). This ring corresponds to a dense accumulation of iron-laden microglia at the edge of the lesion. Instead of clearing debris and promoting repair, these immune cells remain chronically activated. Iron accumulation causes oxidative stress, which is toxic to neurons and oligodendrocytes (myelin-producing cells). This “toxic rim” slowly eats away at the healthy tissue surrounding the lesion. A proportion of PRLs are also slowly expanding lesions (SELs).Because these lesions do not shut down as standard lesions do, they are a primary driver of neurodegeneration (brain shrinkage). The ongoing inflammation at the rim severs nerve fibres or axons. Once an axon is lost, it generally cannot be replaced. Patients with multiple PRLs typically show accelerated brain atrophy (shrinkage), particularly in the deep grey matter, which is strongly correlated with physical and cognitive disability.PRLs are a major biological cause of smouldering MS and PIRA (Progression Independent of Relapse Activity). Traditionally, disability was thought to accumulate via acute relapses (attacks). PRLs cause disability to worsen between attacks. A person with MS may not have a new relapse for years, yet their walking or cognition slowly deteriorates because these smouldering lesions are quietly destroying tissue in the background.PRLs are associated with poor outcomes because they are difficult to treat. The inflammation in a PRL is trapped behind the blood-brain barrier (BBB). Most standard DMTs work by stopping immune cells from entering the brain from the blood. Since the immune cells in a PRL are already inside the brain and self-perpetuating, these drugs are less effective at stopping smouldering inflammation. Tolebrutinib is the first DMT to show an effect in smouldering MS, presumably by targeting PRLs and other intrathecal processes driving smouldering MS, which is why this FDA response is so devastating for people with MS.The FDA are also behind the curve when it comes to smouldering MS and are questing whether or not it is a real entity. I suggest they ask people with MS what smouldering MS is. If they read some of the comments on MS-Selfie, they would recognise that this is clearly the unmet need in treating MS and most people with MS, particularly those with advanced MS who identify as ha...
Transcribe →
Necessary, but insufficient
Jan 1300:04:37Tap to summarizeI keep getting pushback from people, including many colleagues, when I claim that the Epstein-Barr virus (EBV) is the cause of MS. This claim is based on a large body of epidemiological studies over the last 20 years and on applying causation theory. The most important observation supporting my claim is that people who are EBV-seronegative are protected from getting MS. In other words, EBV is necessary to get MS, but is clearly insufficient as not all people who are infected with EBV develop MS, i.e. other factors must contribute to causing MS in addition to EBV.The phrase “necessary but insufficient” describes a condition or factor that must be present for a specific outcome to happen, but cannot make that outcome happen on its own. Think of it as a “required ingredient” rather than a “guarantee.”The Breakdown* Necessary: You absolutely cannot develop MS without EBV. If EBV is missing, MS is impossible.* Insufficient: Having EBV alone does not guarantee you will get MS. Other factors are then required for someone to develop MS.Other examples1. Oxygen and Fire* Necessary: Oxygen is required for a fire to burn. You cannot have fire without it.* Insufficient: Just having oxygen isn’t enough to start a fire. You also need fuel and heat.* Therefore, oxygen is necessary but insufficient for fire.2. A Lottery Ticket* Necessary: You cannot win the lottery if you don’t buy a ticket.* Insufficient: Buying a ticket doesn’t guarantee you will win. You also need luck (the right numbers).3. In a Medical Context (Revalidation)* Necessary: Attending your annual appraisal is necessary to keep your license to practice.* Insufficient: Merely showing up is insufficient to successfully complete your appraisal. You must also demonstrate quality improvement, gather feedback, and show you are up to date.A Simple FormulaFactor A + [Other Factors] = Success* Factor A is “necessary but insufficient.”* If you remove Factor A, the equation fails.* If you only have Factor A, the equation is incomplete.Why is this distinction important?In logic and critical thinking, confusing these two helps avoid errors in judgment. For example, a politician might argue, “We increased police funding, so crime will drop.”A critic might reply: “Funding is necessary for a good police force, but insufficient to solve crime on its own—you also need social programs and economic stability.”EBV-negative MSThe counter-argument a lot of people make is that they have MS, but are EBV-negative. I counter this by saying that they may not be EBV-negative. All assays have false-negative and false-positive rates. To illustrate this in an International Clinically Isolated Syndrome (CIS) study, we found that 41 (3.9%) of 1,047 subjects with CIS were EBV-negative using screening serology. When we tested them with more sensitive assays, only one remained EBV-negative.EBV (true-positive) + [Other Factors] = multiple sclerosisThe other issue is whether we are sure the person who is EBV-negative (true negative) has MS or another disease. We know that at least 1 in 20 people diagnosed with MS in life turn out to have another disease. This is why I think EBV-seronegativity is an important clinical flag and should make one reconsider the diagnosis of MS.I am firmly in the camp that EBV-seronegative MS is likely not to exist, i.e. if you are truly EBV-negative, then you have another disease. Please be aware that the McDonald criteria for diagnosing MS completely ignore this, and there is no mention of EBV serology as a diagnostic aid in helping make the diagnosis of MS or in not making a diagnosis of MS.The futureGetting naysayers to understand and accept “EBV is necessary, but insufficient to develop MS” is important, as they often review and reject grant applications. And this statement underpins using an EBV vaccine to prevent MS. My domino model of MS attempts to illustrate this. Is it clear enough, or do I need to simplify it to illustrate the point? Do any of you have any suggestions to make it easier to grasp?If you are interested in learning more, you can watch my lecture on EBV and MS, which I recently gave at the Nobel Symposium in Stockholm.I would be interested in hearing your thoughts on the concepts in this newsletter. Do you agree with me, or not? If you don’t, can you let me know why?Paper of interestDobson R, Kuhle J, Middeldorp J, Giovannoni G. Epstein-Barr-negative MS: a true phenomenon? Neurol Neuroimmunol Neuroinflamm. 2017 Feb 3;4(2):e318. doi: 10.1212/NXI.0000000000000318.Accidental readersIf you have been forwarded this email and are not an MS-Selfie subscriber, please consider subscribing and helping MS-Selfie expand its resources for the broader MS community. MS-Selfie relies on subscriptions to fund its curated MS-Selfie microsite, MS-Selfie books, MS-Selfie Infocards, and other activities that extend beyond the MS-Selfie Substack newsletters.Subscriptions and donationsMS-Selfie newsletters and access to the MS-Selfie microsite are free. In comparison, off-topic Q&A sessions are restricted to paying subscribers. Subscriptions are being used to run and maintain the MS Selfie microsite and other related activities, as I don’t have time to do this myself. You must be a paying subscriber to ask questions unrelated to the newsletters or podcasts. If you can’t afford to become a paying subscriber, please email a request for a complimentary subscription (ms-selfie@giovannoni.net).QuestionsIf you have questions unrelated to the newsletters or podcasts, please email them to ms-selfie@giovannoni.net. Prof. G will try to answer them as quickly as possible.Important Links📋 MS-Selfie microsite💰 Donations to MS-Selfie👈 Prof. G’s Backstory and CV💾 Prof. G’s MS Blog Archive❓ Conflicts of Interest🦋 BlueSky Social🔗 LinkedIn🖋 MediumGeneral DisclaimerPlease note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Queen Mary University of London or Barts Health NHS Trust. The advice is intended as general and should not be interpreted as personal clinical advice. If you have any problems, please tell your healthcare professional, who can help you. This is a public episode. If you'd like to discuss this with other subscribers or get access to bonus episodes, visit gavingiovannoni.substack.com/subscribe
Transcribe →
New MS subtypes or not?
Jan 500:14:36Tap to summarizeSeveral subscribers have asked me to comment on the new classification system for multiple sclerosis (MS) published last month in the journal Brain, which uses MRI and serum neurofilament levels. One person asked whether this means MS is now two diseases rather than one. Other commentators have hinted that this will change how we diagnose, treat and manage MS.In short, none of these claims are supported by the research findings presented in this paper (Willard et al. Combined magnetic resonance imaging and serum analysis reveals distinct multiple sclerosis types. Brain. 2025 Dec 4;148(12):4578-4591). The article introduces a new machine learning model that integrates MRI data with serum neurofilament light chain (sNfL) levels to supposedly categorise MS based on ‘biology’ rather than ‘MS-related symptoms’. The assumption that MS-related symptoms are not biological is incorrect. The fact that humans, and by inference, people with MS, are biological machines means their symptoms are biological.The Oxford dictionary defines biology as ‘the study of living organisms, divided into many specialised fields that cover their morphology, physiology, anatomy, behaviour, origin, and distribution.’ Symptoms of a disease are hence biological.The study combined fluid biomarkers (sNFL) with brain imaging, which then identified two distinct disease subtypes: early-sNfL and late-sNfL. The early-sNfL group is characterised by higher levels of inflammatory activity, significant lesion accrual, and faster brain atrophy, whereas the late-sNfL group exhibits more gradual neurodegeneration. The authors claim the multimodal approach, including sNFL, proved superior to MRI-only models in correlating with patient disability and predicting how individuals respond to therapeutic interventions. They claim the findings may provide a prognostic framework to support personalised medicine by identifying more aggressive disease states earlier in the clinical course, which are more likely to predict treatment response.LimitationsPlease note that the authors have identified several limitations and flaws in their study, which can be categorised into issues with the study population, methodology, and challenges related to clinical implementation. The study utilised data drawn from clinical trial cohorts, which means the participants do not fully represent the broader MS population. Due to strict eligibility criteria in the source trials, the study lacks data on underrepresented ethnic groups and patients with co-morbidities. While the model was trained on a cohort including both relapsing–remitting and secondary progressive MS, the external validation was conducted only on a cohort of newly diagnosed patients (early MS). Consequently, the model’s accuracy for late-stage disease remains to be validated.The external test dataset had a limited range of EDSS scores, which likely contributed to lower correlation coefficients between the model stages and disability measures in the test set than in the training set. Although the study aimed to use unsupervised machine learning, the pipeline was “not entirely unsupervised” because the initial feature selection step relied on correlations with the EDSS. The researchers noted that the decision to narrow the selection to exactly five variables was an “arbitrary decision” guided by the dataset’s size and resources. The SuStaIn algorithm assumes that disease progression follows a “monotonic sequence,” in which subtypes accumulate abnormalities in a fixed order. This assumption facilitates modelling but may limit the model’s sensitivity to fluctuating disease trajectories. In the longitudinal analysis, sNfL levels decreased in untreated control subjects. The authors attribute this to “regression to the mean,” as patients were recruited during active phases of inflammation (a requirement for trial entry), which naturally subsided, complicating the assessment of actual therapeutic effects.The authors acknowledge that few hospitals currently possess the necessary infrastructure to convert routine MRI scans into the precise quantitative measures required by their model. Quantitative MRI measures are sensitive to differences in scanners and acquisition parameters. While the study used harmonisation (the ComBat algorithm) to mitigate this, such harmonisation poses practical challenges for widespread clinical adoption. The correlations between the model-derived stages and the EDSS were weak. The authors argue this is expected because EDSS is weighted toward motor function, while MRI/sNfL changes often precede clinical symptoms. Still, it highlights a gap between the biological staging and current clinical disability measures. To ensure the model remained accessible for potential clinical translation, the study excluded more advanced imaging modalities (such as myelin-sensitive MRI sequences), which might have provided more comprehensive insights.I am clearly not on the same page as the authors.My initial thoughtsThe title refers to distinct MS subtypes. Use of the term “distinct" is a misnomer, as some subjects switched from one subtype to the other.“Given that 7% of patients switched from one subtype to another in the training dataset, and 23% switched in the testing dataset, these subtypes are likely to represent a continuum of underlying pathology.”The title is therefore misleading and argues against two distinct subtypes of MS.Please note that the training dataset was derived from a phase 2 Evobrutinib clinical trial conducted between March 2017 and July 2018. The study subjects were selected using well-defined inclusion and exclusion criteria. All trial subjects had to have one or more documented relapses within the 2 years before screening, with either one relapse which occurred within the last year before randomisation or the presence of at least one T1 gadolinium-enhancing lesion within 6 months before randomisation—about a quarter of study subjects had been exposed to DMTs in the past.Therefore, the cohort used for training and model development had established active MS, as defined by relapses and/or Gd-enhancing lesions on MRI. Another issue is that they were diagnosed using the 2011 McDonald criteria. This creates problems, as subjects with active MS tend to become less active over time due to regression to the mean. In addition, the consequences of having active MS will then unfold over time as part of the natural history of MS. I would be interested to know how the model would have been developed if it included pwMS who did not have active MS as defined by the trial inclusion criteria. I suspect very differently.Is the model simply measuring baseline MS disease activity, including raised sNFL as an activity marker, and then predicting the consequences of this period of MS disease activity on the end organ, i.e., brain volume loss or end-organ damage? In comparison, those without activity at baseline, i.e. a normal sNFL, will not have the same trajectory in terms of the pathology in the end-organ as measured by MRI, but are likely to regress to the mean in the opposite direction; i.e. have a delayed rise in sNFL as part of the fluctuation in MS disease activity over time. I think any model of MS, a dynamic disease, needs more than a few limited MRI metrics and sNFL to capture its behaviour.In comparison to the training dataset, the testing dataset was from the phase 3 REFLEX trial that compared two dosing frequencies of subcutaneous interferon beta-1a in patients with a first clinical demyelinating event suggestive of multiple sclerosis or CIS. These subjects were diagnosed using the older 2005 McDonald criteria, were younger and were naive to DMTs. The subjects in this study had CIS rather than established MS. I know that a subsequent analysis of the REFLEX clinical trial population, retrospectively applying the McDonald 2017 MS diagnostic criteria, estimated that about 50% of these subjects would have been classified as having MS. This means that 50% didn’t fulfill the requirements for MS and are more likely to have benign MS. Validating the model developed on established active MS using an early, much younger group of subjects naive to DMTs diagnosed with different diagnostic criteria makes little sense to me, i.e. it is flawed from a scientific perspective, and is likely to introduce bias. For example, the validation using subjects very early in the course of their disease, who were much younger and hence had a greater ability to recover function and repair damage, introduces a biological variable not measured by the model. How does neurological reserve and recovery of function impact the model?An essential aspect of clinical outcomes and various biomarkers in MS is that they evolve at different rates. In other words, the temporal sequence of how they change over time is out of sequence with each other. I have referred to this phenomenon in the past as lag. Let me give you some examples. Demyelination develops along a specific pathway before loss of function and before a Gd-enhancing lesion is seen. Changes in the magnetisation transfer ratio (MTR) on MRI can occur in normal-appearing white matter (NAWM) several months before an MS lesion becomes visible with Gd-enhancement. This indicates that MTR changes are earlier and more sensitive markers of pre-lesional tissue damage than Gd enhancement. At some point, axonal injury occurs, leading to NFL release. Based on serial sampling studies, we suspect this process may precede the onset of clinical relapse symptoms and the detection of Gd enhancement on MRI. However, these latter processes are likely to cluster into a relatively narrow window that lasts day...
Transcribe →
Sylvia
Dec 2900:13:38Tap to summarizeSylvia found us, and that is the primary learning point of this case study.Sylvia was diagnosed with MS two months into an internship with an investment bank in London. She probably had an episode of optic neuritis when she was 17, whilst studying for her A-levels. She had transient blurring of vision in her right eye, accompanied by a mild headache behind the eye. Her vision was minimally affected and was back to normal within a week or two. Her general practitioner diagnosed her as having migraine. However, she has the telltale signs of prior optic neuritis when you examine her vision: abnormal colour vision, optic disc pallor, and slowed conduction velocity in that eye on visual evoked potential testing. Sylvia was then well until she developed weakness in her right foot during her final exams at University. She had difficulty lifting her right foot, and it would catch when she walked, causing her to trip several times. In addition, her left leg felt odd with reduced sensation that was more pronounced in the lower leg. As Sylvia was studying for her exams, she put off seeing the campus GP and waited until she returned home. At this point, the weakness had almost resolved. At her mother’s insistence, Sylvia saw the family GP, who advised her to see a specialist.By the time Sylvia saw a neurologist at a central London teaching hospital, the weakness had resolved. However, when the neurologist examined her, he detected persistent weakness in her right foot and some altered sensation in her left foot. He didn’t tell Sylvia what he was thinking, but said she needed an MRI of the brain and spinal cord. Sylvia did not question the neurologist at this stage, as she was excited about her summer ahead. She had a holiday planned with her boyfriend and was due to start a prestigious paid internship at a large international investment bank. Life was good.Two weeks later, she was called by the hospital’s neuroradiology department, saying they had a cancellation and were offering her the slot if she was free. The problem is that she needed to come in that afternoon for her MRI. As Sylvia had yet go on holiday, she was able to postpone seeing her friends that afternoon and attend the hospital for her MRI. Things changed dramatically two weeks later, when she was on holiday in Greece, she received a call from the consultant neurologist saying her MRI had shown some evidence of inflammation in the spinal cord and brain and that he wanted her to have some additional tests. At no point did he mention the cause of the inflammation.A week later, on Monday morning, Sylvia was escorted into the daycase ward at the hospital for blood tests and a lumbar puncture. Sylvia had to wait about an hour as the trainee doctor needed some of the blood results before doing the lumbar puncture. The lumbar puncture was uneventful. She remembers feeling a cold sensation on her lower back, some pressure, and then, three minutes later, the young trainee doctors saying it was all done. Sylvia then had to go to another building that housed the neurophysiology department for electrical tests, including a complete set of evoked potentials and central motor conduction times.You would be wrong to think that Sylvia was a passive recipient of healthcare at this point. Sylvia had immediately Googled spinal cord inflammation, and as a result, she had decided that she likely had multiple sclerosis, and these tests were being done to exclude other conditions and to confirm the diagnosis. Sylvia was a knowledgeable university graduate who had just won one of the most competitive internships in the country. She was an exceptionally bright and capable young lady.To cut a long story short, when Sylvia saw the neurologist three weeks later, she was surprisingly clear-headed and well prepared when he said she had multiple sclerosis. Sylvia had spent the last three weeks reading up on the condition and was ready for what was coming next. He said she had the relapsing-remitting subtype of MS. He said the visual blurring she had had when she was an A-level student was her first attack, which was supported by the abnormal visual evoked potentials showing slow conduction in the right optic nerve.Sylvia wanted to cut to the chase and wanted to ask about treatment. He said she would be eligible for several treatments available on the NHS, but wanted her first to go away and understand what MS was before she could discuss with the MS nurse specific therapies. This was not good enough. Sylvia then begged him for a list of the treatments she could have so she could research them. Reluctantly, he provided her with four options: interferon beta, glatiramer acetate, teriflunomide and dimethyl fumarate. He said that his recommendation would be for dimethyl fumarate. When Sylvia asked him about Tysabri and Lemtrada, he said that she would not be eligible for Tysabri as she had only had one MS attack in the last 12 months and that Lemtrada was too dangerous to use first-line and that he personally did not prescribe alemtuzumab. The issue Sylvia had was that after doing her own research, she had decided she wanted to be treated with Lemtrada or alemtuzumab. Who was this neurologist deciding that alemtuzumab was too dangerous to be used as a first-line treatment? It was at this stage that I received a call from Sylvia’s GP asking if we would be prepared to see Sylvia at the Royal London Hospital for treatment.When I met Sylvia, she was level-headed, intelligent, and street-smart. She knew about all things MS, having spent several hours a day for months reading and learning about MS. She had read our MS Blog and knew all the arguments for and against treating MS aggressively from the start. She had her whole adult life ahead of her and wanted the best chance of living a normal life despite having MS. Contrary to what the neurologist from St Elsewhere had told her, Sylvia fulfilled the NHS criteria for being treated with alemtuzumab, and she went ahead with two courses of treatment in 2015 and 2016.What was not mentioned is that Sylvia had a high lesion load on her MRI and had several gadolinium-enhancing lesions on her initial diagnostic scan, including some subtle enhancement of a lesion in her upper thoracic cord—all the more reason for treating her MS aggressively. From an MS perspective, Sylvia has done well. She has not had any further relapses or new lesions on her MRI, and her brain volume seems to be stable.Sylvia is now married and has two young daughters. She is a senior analyst in the investment bank where she did her internship. She is the primary breadwinner at home. With annual bonuses, she earns over a million pounds per year. The press and media often call upon her to discuss economic issues. She is an ultra-successful young woman, a super-mum. You would not think she had MS.This year marked the tenth anniversary of her MS diagnosis and initial course of alemtuzumab. The question is whether she has been cured of having MS. This is where definitions matter and how we define an MS cure. Although Sylvia has no evident inflammatory MS disease activity (NEIDA), she does have fatigable weakness in her right foot. Sylvia runs half marathons and notices that toward the end of her races and at the end of long training sessions, her right foot begins to drop. Over the last three to four years, the foot drop has started to happen earlier in the race. When Sylvia first began running half-marathons, the foot drop used to occur in the last quarter of the race. She is now noticing it happening from the halfway mark. Is this the first sign of smouldering MS? Or is this simply premature ageing? I don’t know. But if you include ageing or early ageing as part of your definition of smouldering MS, you would say she has smouldering MS. We don’t usually do repeat MRI scans of the spine to monitor MS disease activity. However, I arranged for Sylvia to have an MRI of her spine, and it is clear that the lesion in her upper thoracic spine had gotten longer, and there is evident cord atrophy at the site of the lesion and in the lower cervical cord. So, based on my current thinking of MS, I would say Sylvia has smouldering MS in an existing lesion in the spinal cord. She is aware of this and expects her right foot weakness to worsen gradually. The question Sylvia asks me whenever I see her is: What can be done to slow or stop this process? She is clearly interested in tolebrutinib.I am deliberately using Sylvia as an example of alemtuzumab’s ability to do what it can, i.e., stop new lesion formation and prevent end-organ damage. But what it can’t do is stop smouldering MS that is already established in a small number of lesions before treatment. I have many other examples of people with MS who have no evident inflammatory disease activity nor smouldering associated worsening 15 years or more after being treated with alemtuzumab. I think these people have been cured of having MS. I am aware of similar examples in patients treated with AHSCT. I suspect that in time, we will also see some cures after treatment with cladribine. For a more detailed discussion on defining an MS cure, please see my previous newsletter, ‘To cure or not to cure MS, that is the question’ (24-Aug-2023).I have little doubt that if Sylvia had lived in a different era and had not been promptly treated with alemtuzumab, she would be much worse off than she is now. The counterfactual here would be very different. The only reason Sylvia has done so well and will do well long term is that she is an educated, determined, and self-confident individual who was not prepared to take no for an answer. She knew her rights and made sure she got her MS treated the way she want...
Transcribe →